7 yo male presented with respiratory distress, on and off fever for 4 weeks. Strep test was negative. There had been no improvement with antibiotic treatment. He also had prominent cervical lymphadenopathy.
•Complete Blood Count: WBC: 17.24 x109 /L H; DC: 67.5% blasts, 1.4% neutrophils, 29.1% lymphocytes and 2% monocytes; Hgb: 5.6 g/dL L; HCT: 17.0% L; RBC: 1.74 M/ul; Platelet Ct: 28 K/ ul.
PB smear (Wright Giemsa and cytochemical stains):
•Complete Blood Count: WBC: 17.24 x109 /L H; DC: 67.5% blasts, 1.4% neutrophils, 29.1% lymphocytes and 2% monocytes; Hgb: 5.6 g/dL L; HCT: 17.0% L; RBC: 1.74 M/ul; Platelet Ct: 28 K/ ul.
PB smear (Wright Giemsa and cytochemical stains):
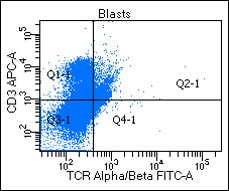
Flow cytometry analysis:
Cytogenetic: 46,XY,del(12)(p12)[20]
Diagnosis: Early T-Cell precursor acute lymphoblastic leukemia (ETP ALL)
•A subset of T-ALL with immunophenotypic and genetic features indicating only limited early T-cell differentiation, with retention of some myeloid and stem cell characteristics.1
•By definition, ETP ALL is positive for CD7, negative for CD1a and CD8, and is positive for at lease one of the myeloid /stem cell markers CD34, CD117, HLA-DR, CD13, CD33, CD11b, or CD65. CD5 is often negative and when positive is present on <75% of the blast population.
• ETP ALL typically also expresses CD2 and cytoplasmic CD3 and may express CD4, but these are not part of the definition.
•Myeloid associated gene mutations, such as FLT3, NRAS/KRAS, DNMT3A, IDH1, and IDH2, are reported at high frequency in ETP ALL. 2-3
•More typical T-ALL–associated mutations such as activating mutations in NOTCH1 or mutations/deletion in CDKN1/2 are infrequent. 4
• Initial studies of ETP ALL suggested that outcome was very poor. 1, 5
•More recent larger series with more effective therapy showed either a small but statistically nonsignificant difference in outcome, or no prognostic significance. 6,7
ETP ALL vs T/myeloid Mixed Phenotypic Acute Leukemia (MPAL)
•The expression of one or multiple myeloid markers in ETP ALL often raises the possibility of diagnosis for T/Myeloid MPAL.
•Although ETP ALL is a kind of “T/myeloid” leukemia with many myeloid genetic features, it is practically significant to differentiate ETP ALL and T/myeloid MPAL since they are typically treated differently.
•From a definitional perspective, WHO criterial for myeloid lineage assignment (as shown in the table below) should be applied.
•In this case, there was no MPO expression based on flow cytometry and cytochemical stain. The blasts were negative for lysozyme, CD14, CD11c, nonspecific esterase, so they do not meet the criteria for monocytic differentiation.
Please be aware that the following criteria for lineage assignment do not universally apply for the diagnosis of AML or ALL. When MPAL is not being considered, cases of AML or ALL do not need to meet more strict MPAL criteria in order to assign lineage. 8
Diagnosis: Early T-Cell precursor acute lymphoblastic leukemia (ETP ALL)
•A subset of T-ALL with immunophenotypic and genetic features indicating only limited early T-cell differentiation, with retention of some myeloid and stem cell characteristics.1
•By definition, ETP ALL is positive for CD7, negative for CD1a and CD8, and is positive for at lease one of the myeloid /stem cell markers CD34, CD117, HLA-DR, CD13, CD33, CD11b, or CD65. CD5 is often negative and when positive is present on <75% of the blast population.
• ETP ALL typically also expresses CD2 and cytoplasmic CD3 and may express CD4, but these are not part of the definition.
•Myeloid associated gene mutations, such as FLT3, NRAS/KRAS, DNMT3A, IDH1, and IDH2, are reported at high frequency in ETP ALL. 2-3
•More typical T-ALL–associated mutations such as activating mutations in NOTCH1 or mutations/deletion in CDKN1/2 are infrequent. 4
• Initial studies of ETP ALL suggested that outcome was very poor. 1, 5
•More recent larger series with more effective therapy showed either a small but statistically nonsignificant difference in outcome, or no prognostic significance. 6,7
ETP ALL vs T/myeloid Mixed Phenotypic Acute Leukemia (MPAL)
•The expression of one or multiple myeloid markers in ETP ALL often raises the possibility of diagnosis for T/Myeloid MPAL.
•Although ETP ALL is a kind of “T/myeloid” leukemia with many myeloid genetic features, it is practically significant to differentiate ETP ALL and T/myeloid MPAL since they are typically treated differently.
•From a definitional perspective, WHO criterial for myeloid lineage assignment (as shown in the table below) should be applied.
•In this case, there was no MPO expression based on flow cytometry and cytochemical stain. The blasts were negative for lysozyme, CD14, CD11c, nonspecific esterase, so they do not meet the criteria for monocytic differentiation.
Please be aware that the following criteria for lineage assignment do not universally apply for the diagnosis of AML or ALL. When MPAL is not being considered, cases of AML or ALL do not need to meet more strict MPAL criteria in order to assign lineage. 8

References:
1. Coustan-Smith E, Mullighan CG, Onciu M, et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol. 2009;10(2):147-156.
2. Neumann M, Heesch S, Schlee C, et al. Whole exome sequencing in adult ETP-ALL reveals a high rate of DNMT3A mutations. Blood. 2013; 121(23):4749-4752.
3. Neumann M, Coskun E, Fransecky L, et al. FLT3 mutations in early T-cell precursor ALL characterize a stem cell like leukemia and imply the clinical use of tyrosine kinase inhibitors. PLoS One. 2013;8(1):e53190.
4. Zhang J, Ding L, Holmfeldt L, et al. The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature. 2012; 481(7380):157-163.
5. Inukai T, Kiyokawa N, Campana D, et al. Clinical significance of early T-cell precursor acute lymphoblastic leukaemia: results of the Tokyo Children’s Cancer Study Group Study L99-15. Br J Haematol. 2012;156(3):358-365.
6. Patrick K, Wade R, Goulden N, et al. Outcome for children and young people with Early T-cell precursor acute lymphoblastic leukaemia treated on a contemporary protocol, UKALL 2003. Br J Haematol. 2014; 166(3):421-424.
7. Wood BL, Winter S, Dunsmore KP, et al. T-lymphoblastic leukemia (T-ALL) shows excellent outcome, lack of significance of the early thymic precursor (ETP) immunophenotype, and validation of the prognostic value of end-induction minimal residual disease (MRD) in Children’s Oncology Group (COG) Study AALL0434 [abstract]. Blood. 2014;124(21):1.
8. Arber DA, Orazi A, Hasserjian, R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016; 127(20): 2391-2405.
9. Borowitz, Michael J . Mixed phenotype acute leukemia. Cytometry Part B: Clinical Cytometry. 2014; 86(3): 152-153
1. Coustan-Smith E, Mullighan CG, Onciu M, et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol. 2009;10(2):147-156.
2. Neumann M, Heesch S, Schlee C, et al. Whole exome sequencing in adult ETP-ALL reveals a high rate of DNMT3A mutations. Blood. 2013; 121(23):4749-4752.
3. Neumann M, Coskun E, Fransecky L, et al. FLT3 mutations in early T-cell precursor ALL characterize a stem cell like leukemia and imply the clinical use of tyrosine kinase inhibitors. PLoS One. 2013;8(1):e53190.
4. Zhang J, Ding L, Holmfeldt L, et al. The genetic basis of early T-cell precursor acute lymphoblastic leukaemia. Nature. 2012; 481(7380):157-163.
5. Inukai T, Kiyokawa N, Campana D, et al. Clinical significance of early T-cell precursor acute lymphoblastic leukaemia: results of the Tokyo Children’s Cancer Study Group Study L99-15. Br J Haematol. 2012;156(3):358-365.
6. Patrick K, Wade R, Goulden N, et al. Outcome for children and young people with Early T-cell precursor acute lymphoblastic leukaemia treated on a contemporary protocol, UKALL 2003. Br J Haematol. 2014; 166(3):421-424.
7. Wood BL, Winter S, Dunsmore KP, et al. T-lymphoblastic leukemia (T-ALL) shows excellent outcome, lack of significance of the early thymic precursor (ETP) immunophenotype, and validation of the prognostic value of end-induction minimal residual disease (MRD) in Children’s Oncology Group (COG) Study AALL0434 [abstract]. Blood. 2014;124(21):1.
8. Arber DA, Orazi A, Hasserjian, R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016; 127(20): 2391-2405.
9. Borowitz, Michael J . Mixed phenotype acute leukemia. Cytometry Part B: Clinical Cytometry. 2014; 86(3): 152-153